BOOK OF ABSTRACTS | HEALTH EQUITY
BOOKS OF ABSTRACTS | HEALTH EQUITY
Brief Research Article | HIV and AIDS
COMMENTARY | COVID-19 AND HIV/AIDS
COMMENTARY | COVID-19 VACCINE
COMMENTARY | DIGITAL HEALTH
COMMENTARY | EMERGENCY PRACTICE
COMMENTARY | MATERNAL MORTALITY
COMMENTARY | PERINATAL HIV TRANSMISSION
LETTER TO THE EDITOR | COVID-19 PANDEMIC
LETTER TO THE EDITOR | COVID-19 TEST
METHODOLOGY ARTICLE | HIV
NOTES FROM THE FIELD | COVID-19 PANDEMIC
NOTES FROM THE FIELD | MCH STUDENT ASSOCIATION
ORIGINAL ARTICLE | ADOLESCENT HEALTH
ORIGINAL ARTICLE | BIRTH INTERVAL
ORIGINAL ARTICLE | CAFFEINE AND CHILDHOOD OBESITY
ORIGINAL ARTICLE | CHILD HEALTH
ORIGINAL ARTICLE | CLUBFOOT
ORIGINAL ARTICLE | COMPLEMENTARY FEEDING PRACTICES
ORIGINAL ARTICLE | CONTINUUM OF CARE
ORIGINAL ARTICLE | COVID-19 DISEASE
ORIGINAL ARTICLE | COVID-19 IN PREGNANCY
ORIGINAL ARTICLE | COVID-19 OUTCOMES
ORIGINAL ARTICLE | DEPRESSION & HIV
ORIGINAL ARTICLE | DIETARY MICRONUTRIENTS AND HIV
ORIGINAL ARTICLE | HIV AND PREGNANT WOMEN
ORIGINAL ARTICLE | HIV INFECTION
ORIGINAL ARTICLE | HIV PREVENTION
ORIGINAL ARTICLE | HIV SCREENING
ORIGINAL ARTICLE | HIV TESTING
ORIGINAL ARTICLE | HIV Viral Load
ORIGINAL ARTICLE | HIV-TB CO-INFECTIONS
ORIGINAL ARTICLE | HIV/AIDS
ORIGINAL ARTICLE | HIV/AIDS IN ECUADOR
ORIGINAL ARTICLE | HOME DELIVERY
ORIGINAL ARTICLE | HOUSING AND HOUSING INEQUALITIES
ORIGINAL ARTICLE | IMMUNIZATION
ORIGINAL ARTICLE | INFLAMMATORY BOWEL DISEASE
ORIGINAL ARTICLE | LEISHMANIASIS
ORIGINAL ARTICLE | LIFE EXPECTANCY
ORIGINAL ARTICLE | MALARIA & HIV
ORIGINAL ARTICLE | MATERNAL HEALTH
ORIGINAL ARTICLE | MATERNAL MORTALITY
ORIGINAL ARTICLE | MATERNAL OBESITY
ORIGINAL ARTICLE | MORTALITY
ORIGINAL ARTICLE | NEONATAL MORTALITY
ORIGINAL ARTICLE | OBSTETRIC FISTULA REPAIR
ORIGINAL ARTICLE | OXYTOCIN VS MISOPROSTOL IN PPH
ORIGINAL ARTICLE | PEDIATRIC HIV
ORIGINAL ARTICLE | PREGNANCY
ORIGINAL ARTICLE | PRENATAL CARE
ORIGINAL ARTICLE | PUERPERAL SEPSIS
ORIGINAL ARTICLE | REPRODUCTIVE HEALTH
ORIGINAL ARTICLE | TEENAGE PREGNANCY
ORIGINAL ARTICLE | VACCINATION
ORIGINAL ARTICLE | VACCINE
ORIGINAL ARTICLE | VACCINE EQUITY
ORIGINAL ARTICLE | VIRTUAL PRENATAL CARE
ORIGINAL ARTICLE | WOMEN HEALTH
ORIGINAL ARTICLE | WOMEN’S HEALTH
ORIGINAL ARTICLE | YOUTH MORTALITY
Original Research | Article Healthcare
ORIGINAL RESEARCH ARTICLE | PEDIATRIC HIV
PUBLIC HEALTH PRACTICE | ACADEMIC DETAILING
PUBLIC HEALTH PRACTICE | CHILD DEVELOPMENT
PUBLIC HEALTH PRACTICE | CHILD HEALTH
PUBLIC HEALTH PRACTICE | HIV
PUBLIC HEALTH PRACTICE | OPT-OUT APPROACH IN HIV TESTING
PUBLIC HEALTH PRACTICE | PREGNANCY WHEEL
PUBLIC HEALTH PRACTICE | SURVEILLANCE
SHORT RESEARCH COMMUNICATION
SHORT RESEARCH COMMUNICATION | COVID AND MENTAL HEALTH
SHORT RESEARCH COMMUNICATION | COVID MCH RESEARCH AGENDA
SHORT RESEARCH COMMUNICATION | COVID-19
SHORT RESEARCH COMMUNICATION | COVID-19 AND CHILD VACCINATION
SHORT RESEARCH COMMUNICATION | COVID-19 AND MATERNAL MORTALITY
SHORT RESEARCH COMMUNICATION | COVID-19 AND REMOTE WORKERS
SHORT RESEARCH COMMUNICATION | COVID-19 PANDEMIC DISPARITIES
SHORT RESEARCH COMMUNICATION | HEALTHCARE PROVIDER TRAINING
SHORT RESEARCH COMMUNICATION | MALARIA
SHORT RESEARCH COMMUNICATION | SINGLETONS, TWINS, MULTIPLE BIRTHS
SHORT RESEARCH COMMUNICATION | SURVEILLANCE SYSTEM FOR COVID-19




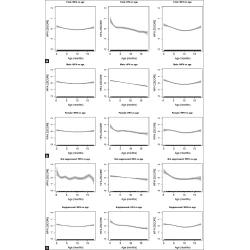





COMMENTARY | MATERNAL MORTALITY
Thirty Years of United Nations Inter-Agency Working Group’s Global, Regional, and National Maternal Mortality Estimates Revisited
Yifru Berhan, Sinteyehu Abeba
DOI: 10.25259/IJMA_679
Full text |
|  PDF
PDF 